Why study cattle reproduction?
|
In agriculture, infertility affects intensive food production, where conception rates to artificial insemination in dairy cows have decreased by approximately 1% per year over recent decades [1]. Despite most inseminations resulting in successful fertilisation, embryonic and foetal mortality occurs in about 50% of the cases, with an estimated 70–80% of losses sustained before the embryo attaches to the uterus (a process called implantation) [2]. According to the 2019 Eurostat report, milk represents the highest share of the EU-28’s agricultural industry, accounting for 13.2% of the total agricultural output value, whereas beef represents 7.7% of total agricultural output [3]. Thus, together with the need to increase our food supply to meet demands of a rapidly growing population, reproductive inefficiency has a tremendous social and economic impact for the EU.
|
How can paternal factors affect pregnancy success?
Many factors are involved in implantation failure, but in several species, there is an increased interest in the role of the maternal immune system and its regulation by the components of the male ejaculate, in particular seminal plasma. Seminal plasma is a mixture of fluids excreted from the testes, epididymis and accessory glands containing sugars, salts, lipids, proteins, and various other factors. In mice and pigs, exposure of the female reproductive tract to seminal plasma leads to increased embryo viability and improves their ability to implantate [4, 5]. These positive effects are thought to be due to the ability of seminal plasma to regulate the maternal immune system, inducing immune tolerance towards paternal antigens during pregnancy [6, 7]. This is important because the embryo exhibits both maternal and paternal antigens. If immune tolerance towards these paternal antigens did not take place, the immune system would recognise them as non-self and destroy the embryo, resulting in pregnancy loss.
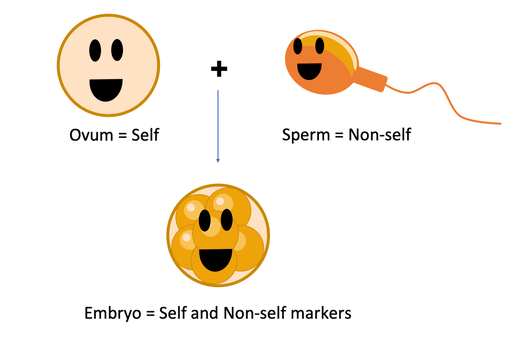
Because an embryo is formed by the union of a cell coming from the mother (the ovum) and a cell coming from the father (the spermatozoa), it will express both maternal (self) and paternal (non-self markers). The latter can be recognised by the maternal immune system and trigger an attack if immune tolerance is not induced. There is evidence in mice that seminal plasma can play a role in the stimulation of maternal immune tolerance towards the embryo.
What is the aim of this project?
Taken collectively, the literature suggests that although seminal plasma is not essential for pregnancy success, it increases the
chances of embryo development and survival through modulation of the maternal environment. However, in cattle, a species in which the dilution or removal of seminal plasma before artificial insemination is routine, evidence for a major role of this fluid in fertility is relatively weak. Due to characteristics of mating in rodents and pigs (species in which the regulatory properties of seminal plasma have been described), seminal plasma reaches the uterus and can therefore interact directly with the endometrium. It is not clear, however, whether any seminal plasma reaches the uterus in species that ejaculate inside the vagina, such as cattle or humans. It is possible that in those species, seminal plasma has an indirect effect on the uterine environment and/or that sperm act as vehicles for the transport of seminal plasma components to far regions of the reproductive tract. In this sense, the bovine model could be more appropriate than rodents or pigs in understanding the regulatory properties of seminal plasma in the maternal environment of women.
Thus, this project aims to understand how immune modulation of the maternal reproductive tract induced by paternal factors (seminal plasma and/or sperm) contributes to pregnancy success in cattle by better understanding the consequences of the changes induced as it pertains to embryo development. If the influence of seminal plasma on embryo survival in mice is recapitulated in cattle, the associated increased growth, production, and efficiency could have a tremendous impact in the beef and dairy industries.
chances of embryo development and survival through modulation of the maternal environment. However, in cattle, a species in which the dilution or removal of seminal plasma before artificial insemination is routine, evidence for a major role of this fluid in fertility is relatively weak. Due to characteristics of mating in rodents and pigs (species in which the regulatory properties of seminal plasma have been described), seminal plasma reaches the uterus and can therefore interact directly with the endometrium. It is not clear, however, whether any seminal plasma reaches the uterus in species that ejaculate inside the vagina, such as cattle or humans. It is possible that in those species, seminal plasma has an indirect effect on the uterine environment and/or that sperm act as vehicles for the transport of seminal plasma components to far regions of the reproductive tract. In this sense, the bovine model could be more appropriate than rodents or pigs in understanding the regulatory properties of seminal plasma in the maternal environment of women.
Thus, this project aims to understand how immune modulation of the maternal reproductive tract induced by paternal factors (seminal plasma and/or sperm) contributes to pregnancy success in cattle by better understanding the consequences of the changes induced as it pertains to embryo development. If the influence of seminal plasma on embryo survival in mice is recapitulated in cattle, the associated increased growth, production, and efficiency could have a tremendous impact in the beef and dairy industries.
References:
[1] Lucy MC. Reproductive loss in high-producing dairy cattle: where will it end? J Dairy Sci, 2001. 84(6): p. 1277-93.
[2] Diskin MG, Waters SM, Parr MH, Kenny DA. Pregnancy losses in cattle: potential for improvement. Reprod Fertil Dev 2016; 28:83–93
[3] Statistical Office of the European Communities. Agricultural accounts and prices. Eurostat Agriculture, forestry and fishery statistics 2019; 70-71.
[4] Bromfield JJ, Schjenken JE, Chin PY, Care AS, Jasper MJ, Robertson SA. Maternal tract factors contribute to paternal seminal fluid impact on metabolic phenotype in offspring. Proc Natl Acad Sci 2014; 111:2200–2205
[5] O’Leary S, Jasper MJ, Warnes GM, Armstrong DT, Robertson SA. Seminal plasma regulates endometrial cytokine expression, leukocyte recruitment and embryo development in the pig. Reproduction 2004; 128:237–247
[6] Guerin LR, Moldenhauer LM, Prins JR, Bromfield JJ, Hayball JD, Robertson SA. Seminal fluid regulates accumulation of FOXP3+ regulatory T cells in the preimplantation mouse uterus through expanding the FOXP3+ cell pool and CCL19-mediated recruitment. Biol Reprod 2011; 85:397–408
[7] Shima T, Inada K, Nakashima A, Ushijima A, Ito M, Yoshino O, Saito S. Paternal antigen-specific proliferating regulatory T cells are increased in uterine draining lymph nodes just before implantation and in pregnant uterus just after implantation by seminal plasma-priming in allogeneic mouse pregnancy. J Reprod Immunol 2015; 108:72–82
[1] Lucy MC. Reproductive loss in high-producing dairy cattle: where will it end? J Dairy Sci, 2001. 84(6): p. 1277-93.
[2] Diskin MG, Waters SM, Parr MH, Kenny DA. Pregnancy losses in cattle: potential for improvement. Reprod Fertil Dev 2016; 28:83–93
[3] Statistical Office of the European Communities. Agricultural accounts and prices. Eurostat Agriculture, forestry and fishery statistics 2019; 70-71.
[4] Bromfield JJ, Schjenken JE, Chin PY, Care AS, Jasper MJ, Robertson SA. Maternal tract factors contribute to paternal seminal fluid impact on metabolic phenotype in offspring. Proc Natl Acad Sci 2014; 111:2200–2205
[5] O’Leary S, Jasper MJ, Warnes GM, Armstrong DT, Robertson SA. Seminal plasma regulates endometrial cytokine expression, leukocyte recruitment and embryo development in the pig. Reproduction 2004; 128:237–247
[6] Guerin LR, Moldenhauer LM, Prins JR, Bromfield JJ, Hayball JD, Robertson SA. Seminal fluid regulates accumulation of FOXP3+ regulatory T cells in the preimplantation mouse uterus through expanding the FOXP3+ cell pool and CCL19-mediated recruitment. Biol Reprod 2011; 85:397–408
[7] Shima T, Inada K, Nakashima A, Ushijima A, Ito M, Yoshino O, Saito S. Paternal antigen-specific proliferating regulatory T cells are increased in uterine draining lymph nodes just before implantation and in pregnant uterus just after implantation by seminal plasma-priming in allogeneic mouse pregnancy. J Reprod Immunol 2015; 108:72–82
This work was funded by the European Union's Horizon 2020 research and innovation programme under grant agreement no. 792212


